In the global faucet and plumbing industry, the term lead-free brass is now widely used. Regulations such as the U.S. Safe Drinking Water Act, NSF/ANSI 372, and the European 4MSI Positive List have pushed manufacturers to significantly reduce lead content in materials that come into contact with drinking water.
However, from a metallurgical perspective, there is a critical fact that is often overlooked:
Brass cannot be completely lead-free in the strict sense.
Even modern lead-free brass faucet materials usually contain trace amounts of lead, typically below regulatory limits. More importantly, the way lead behaves inside a brass alloy microstructure creates challenges that cannot be solved simply by lowering the percentage.
To understand why, we need to look deeper into how brass actually works as a material.
Why Lead Was Added to Brass in the First Place
Brass is an alloy primarily composed of copper and zinc. It has been widely used in plumbing components and faucet manufacturing for more than a century due to its excellent balance of properties:
- Good corrosion resistance
- Strong mechanical performance
- Excellent machinability
- Mature global manufacturing supply chains
The key historical reason for adding lead is machinability.
In machining processes such as drilling, milling, and turning, lead forms tiny dispersed particles inside the metal. These particles act as internal lubricants that help cutting tools break chips efficiently and maintain smooth surface finishes.
This characteristic allows manufacturers to produce complex components such as:
- Faucet bodies
- Valve housings
- Plumbing fittings
with high efficiency and consistent quality.
Without lead, machining brass components becomes significantly more difficult, increasing tool wear, machining time, and production costs.
Where Lead Exists Inside Brass
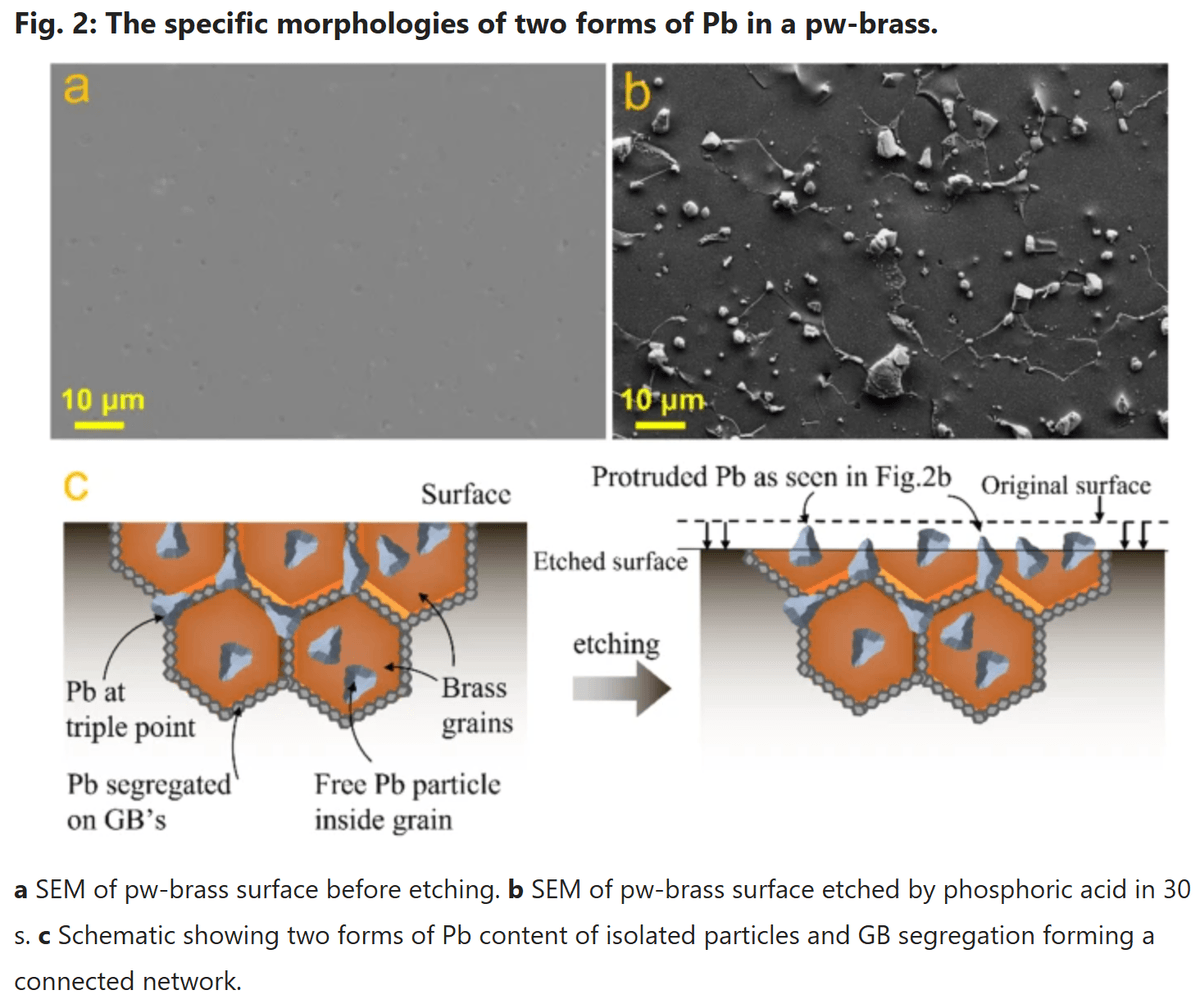
One of the most important metallurgical characteristics of brass is that lead does not dissolve uniformly in the copper-zinc matrix.
Instead, lead typically exists in the microstructure as:
- Discrete microscopic particles
- Inclusions trapped between metal grains
- Small accumulations along grain boundaries
These particles are extremely small and normally remain locked inside the alloy structure.
Under normal conditions, they do not directly interact with water flowing through plumbing systems.
However, certain thermal processes during manufacturing can change how lead behaves within the metal.
How Heating Can Change Lead Distribution
Many plumbing and faucet components go through high-temperature processes during manufacturing or assembly. These may include:
- Brazing
- Soldering
- High-temperature joining operations
When brass is heated to elevated temperatures, several metallurgical changes occur:
- Lead becomes more mobile within the microstructure
- It may migrate toward grain boundaries
- In some cases, it can diffuse toward the surface of the metal
At sufficiently high temperatures, lead can spread along grain boundaries and form extremely thin layers close to the surface.
These changes are microscopic and invisible externally, but they become important once the component is exposed to water.
How Lead Leaching Can Begin
When water contacts a brass surface where trace lead has reached the outer layer, chemical reactions can occur.
The exposed lead quickly oxidizes, forming microscopic compounds on the metal surface. Under certain conditions, these oxidation products can appear as tiny needle-like structures.
Because these particles are loosely attached to the surface, they can be easily washed away by flowing water.
This is the mechanism commonly referred to as lead leaching in plumbing materials.
Importantly, this process typically occurs at the surface level, rather than involving large amounts of lead dissolving from the bulk material.
The Built-In Contradiction of Brass
This leads to a fundamental contradiction in brass alloy design.
The same element that improves machinability can also contribute to the possibility of lead exposure.
In simple terms:
- Lead makes brass easy to manufacture
- But lead also creates microstructural pathways for leaching
Reducing the lead percentage helps significantly, but it does not completely eliminate the underlying metallurgical behavior of the alloy.
That is why modern regulations focus not only on composition limits but also on performance-based testing for drinking water safety.
Examples include:
- NSF/ANSI 61 leaching tests
- NSF/ANSI 372 lead content limits
- The European 4MSI Positive List material requirements (DWD)
How Faucet Manufacturers Reduce the Risk
The faucet and plumbing industry has developed several strategies to minimize potential lead exposure while maintaining the advantages of brass.
Ultra-Low-Lead Brass Alloys
Modern lead-free brass alloys reduce lead content to extremely low levels while maintaining machinability.
However, these alloys often require more advanced machining processes.
Improved Surface Finishing
Surface treatments can help stabilize the outer metal layer and reduce exposure risks. These processes may include:
- Surface polishing
- Chemical passivation
- Controlled finishing processes
Better Manufacturing Control
Manufacturers are increasingly paying attention to temperature control and assembly processes that could influence lead migration.
Process control is now an important factor in producing safe plumbing materials for drinking water systems.
Exploring Alternative Materials
In recent years, many companies have begun exploring materials that eliminate lead entirely.
Examples include:
- Stainless steel faucets
- Advanced copper alloys
- Polymer-based plumbing components
Each alternative has its own engineering challenges. For example, stainless steel provides zero-lead water contact surfaces, but it requires different machining technologies and surface finishing processes compared with brass.
FAQ: Lead-Free Brass in Plumbing Materials
Is lead-free brass really lead-free?
No material labeled as lead-free brass is completely free of lead in the strict metallurgical sense. Instead, it means the lead content is below regulatory limits such as those defined by NSF/ANSI 372.
Why is brass still widely used in faucets?
Brass offers an excellent balance of corrosion resistance, durability, machinability, and cost efficiency, making it one of the most practical materials for faucet manufacturing.
Are stainless steel faucets safer?
Stainless steel contains no lead, which eliminates lead leaching concerns. However, stainless steel introduces different engineering and manufacturing challenges.
Looking Ahead: The Future of Faucet Materials
Brass has been the backbone of the plumbing and faucet industry for decades. Its reliability, durability, and manufacturability are difficult to replace.
However, evolving regulations and growing awareness of drinking water safety are pushing the industry to explore new material platforms.
The key question facing manufacturers today is:
Should we continue optimizing brass alloys, or transition toward entirely new materials?
The answer will likely involve both approaches.
Faucet Materials Series
This article is Part 1 of our technical series on faucet materials and drinking water safety.
Upcoming articles:
Part 2 — DZR Brass vs Low-Lead Brass: What’s the Real Difference?
Part 3 — Stainless Steel Faucets: Trend, Regulation, or Marketing?
Part 4 — What the 4MSI Positive List Means for Faucet Manufacturers
Reference:
Metallurgical pathways of lead leaching from brass | npj Materials Degradation
https://www.nature.com/articles/s41529-023-00389-8